Botulinum Toxin Injections
Botulinum toxin injections (BTX), often referred to by the product names BOTOX® or DYSPORT®, are biological toxins (botulinum toxin) transformed into a therapeutic agent. Work with BTX began in the late 1960s to treat neurological disorders. Today, BTX is used for the treatment of frown lines, forehead furrows, "crow's feet," lines and wrinkles of the lower face, and even nasal muscles (to decrease nostril flaring). BTX injections have proven to be a very popular nonsurgical cosmetic procedure. Aesthetic plastic surgeons have found that the type of lines and wrinkles that respond to BTX injections are those caused by muscles-specifically those muscles that contract during facial expressions such as frowning or squinting.
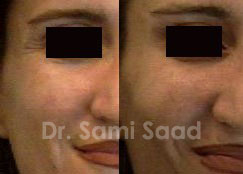
Technique:
The patient is asked to contract the muscles in the area being treated so the surgeon can determine the proper location for injection. In most cases, BTX is injected directly into the muscle with a tiny needle. It takes a few days to realize the effect of BTX injections.
Benefits:
- No downtime. Patients can immediately resume normal activities.
- Long experience has proven BTX to be safe. To date, no systemic complications associated with BTX injections have been documented.
- Treatment is reversible within several months.
- May be beneficial for treatment of migraine headaches.
Other Considerations:
- BTX injections are a temporary solution for the treatment of wrinkles. The effects last from 3-4 months and require repeat treatments.
- Possible side effects include local numbness, swelling, bruising, or a burning sensation during injection. Some patients have reported temporary headache and nausea. Most complications are of short duration and can be avoided with proper injection techniques.
- A small percentage of patients are reported to experience no improvement at all.
Contraindications:
Pregnant or nursing women should postpone undergoing this procedure. It is not known whether injection of BTX has any effect on a fetus or whether it is found in breast milk, although annecdotal reports that BTX has been occasionally injected for women who did not know of their pregnancy, with no evidense of adverse effects..